Seawater General Corrosion & Erosion
Titanium Alloy Mill Product Capabilities
September 26, 2016Commercial Titanium Alloys
September 26, 2016Seawater General Corrosion & Erosion
Ti-Material-Analysis
GRADE 5 (R56400) Ti-6Al-4V
GRADE 1 (R50250)
Grade2 & Grade3
GRADE 4 (R50700)
Grade7 & Grade11
Ti - 0.15Pd (Grade 16)
Ti - 0.1Ru (Grade 27)
Grade9 & Grade12
Ti - 3Al - 2.5V - 0.05Pd
Grade6 & Grade28
Ti - 5Al - 2.5Sn - ELI
Ti-8Al-1Mo-1V & Ti-6Al-2Sn-4Zr-2Mo-0.1Si
Grade23 & Grade29
Ti - 6Al - 6V - 2Sn
Ti-6Al-2Sn-4Zr-6Mo & Ti-6Al-2Sn-2Zr-2Mo-2Cr-0.1
Ti - 5Al - 2Zr - 2Sn - 4Mo - 4Cr
Ti-4.5Al-3V-2Mo-2Fe & Ti-4Al-4Mo-2Sn-0.5Si
Ti - 10V - 2Fe - 3Al
Grade19; Grade20
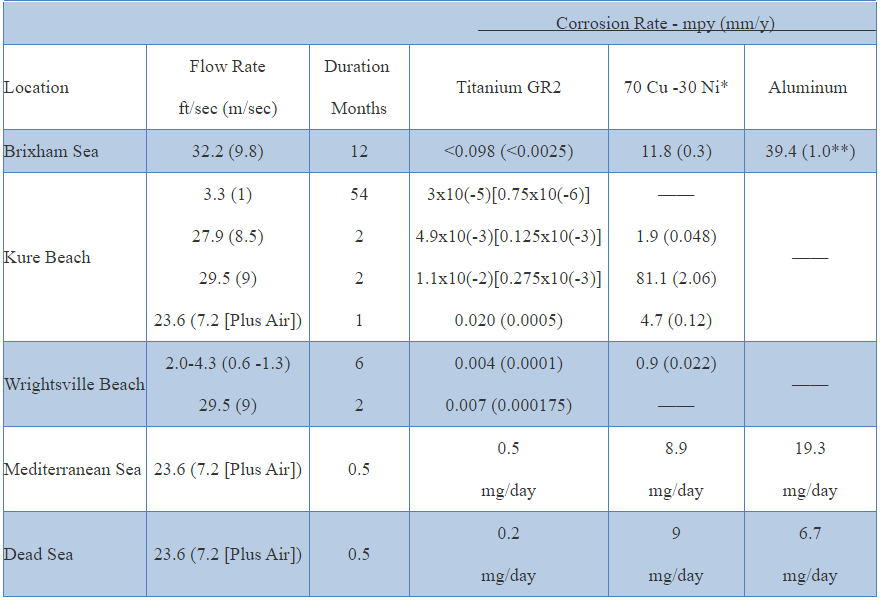
Seawater General Corrosion
Titanium resists corrosion from seawater to temperatures as high as 500°F (260°C). Titanium tubing that is exposed for 16 years to polluted seawater in a surface condenser was slightly discolored but showed no evidence of corrosion. Titanium has provided over thirty years of trouble-free seawater service for the chemical, oil refining, and desalination industries. Exposure of titanium for many years to depths of over a mile below the ocean surface has not produced any measurable corrosion. Pitting and crevice corrosion are totally absent, even if marine deposits form. The presence of sulfides in seawater does not affect the resistance of titanium to corrosion. Exposure of titanium to marine atmospheres or splash or tide zone does not cause corrosion.Erosion
Titanium has the ability to resist erosion by high velocity seawater. Velocities as high as 120 ft./sec. cause only a minimal rise in erosion rate. The presence of abrasive particles, such as sand, has only a small effect on the corrosion resistance of titanium under conditions that are extremely detrimental to copper and aluminum base alloys. Titanium is considered one of the best cavitation-resistant materials available for seawater service.Oxidizing Acids
Titanium is highly resistant to oxidizing acids over a wide range of concentrations and temperatures. Common acids in this category include nitric, chromic, perchloric, and hypochlorous (wet Cl2) acids. These oxidizing compounds assure oxide film stability. Low but finite corrosion rates from continued surface oxidation may be observed under high temperatures & highly oxidizing conditions. Titanium has been extensively utilized for handling and producing nitric acid in applications where stainless steels have exhibited significant uniform or inter-granular attack. Titanium offers excellent resistance over the full concentration range at sub-boiling temperatures. At higher temperatures, however, titanium’s corrosion resistance is highly dependent on nitric acid purity. In hot and very pure solutions, vapor condensates nitric acid and significant general corrosion (and trickling acid condensate attack) may occur in the 20 to 70 wt.% range. Under marginal high temperature conditions, higher purity unalloyed grades of titanium (i.e., Grade 1) are preferred for curtailing accelerated corrosion of weldments.On the other hand, various metallic species such as Si, Cr, Fe, Ti or various precious metal ions (i.e., Pt, Ru) in very minute amounts tend to inhibit high temperature corrosion of titanium in nitric acid solutions. Titanium often exhibits superior performance to stainless steel alloys in high temperatures. Titanium’s own corrosion product Ti+4, is a very potent inhibitor. This is particularly useful in recirculating nitric acid process streams such as stripper re-boiler loops where effective inhibition results from achievement of steady-state levels of dissolved Ti+4. Titanium also offers good resistance to nitric acid vapors. However, titanium is not recommended for use in red fuming nitric acid because of the danger of pyrophoric reactions.
Red Fuming Nitric Acid
Although titanium in general has excellent resistance to nitric acid over a wide range of concentrations and temperatures, it should not be used with red fuming nitric acid. A pyrophoric reaction product can be produced resulting in serious accidents. An investigation of these accidents has shown that the pyrophoric reaction is always preceded by a rapid corrosive attack on titanium. This attack is inter-granular and results in a surface residue of finely divided particles of metallic titanium. These are highly pyrophoric and are capable of detonating in the presence of strong oxidizing agents such as fuming nitric acid. It has been established that the water content of the solution must be less than 1.34% and the NO2 content greater than 6% for the pyrophoric reaction to develop.Chromic Acid
The data on chromic acid is not as extensive as it is on nitric acid. However, corrosion resistance of titanium to chromic acid appears to be very similar to that observed in nitric acid.Reducing Acids
Titanium offers moderate resistance to reducing acids such as hydrochloric, sulfuric, and phosphoric. Corrosion rates increase with increasing acid concentration and temperature. The grade 7 alloy offers the best resistance to these environments followed by grade 12, and non-alloyed titanium and grade 5.Hydrochloric Acid
Iso-corrosion data illustrates that grade 2 offers useful corrosion resistance to about 7% hydrochloric acid at room temperature; grade 12 to about 9% HCl; and grade 7 to about 27%. This resistance is significantly lowered at near boiling temperatures. Small amounts of certain multi-valent metal ions in solutions such as ferric ion can effectively inhibit the corrosion of titanium in hydrochloric acid. When sufficient ferric ion is present, grade 2, 7 and 12 show similar corrosion resistance. Other metal ions such as Cu+2, Ni+2, Mo+6, and Ti+4, also passivate titanium against attack by hydrochloric acid. Oxidizing agents such as nitric acid, chlorine, sodium hypochlorite, or chromate ions, also have been shown to be effective inhibitors. These have allowed titanium to be successfully utilized in many hydrochloric acid applications. Severe corrosion damage on titanium equipment has resulted from cleaning procedures utilizing pure hydrochloric acid or acid inhibited with amines. If hydrochloric or sulfuric acid is used to clean titanium surfaces, it is recommended that sufficient ferric chloride be added to effectively inhibit corrosion of the titanium.Sulfuric Acid
Titanium is resistant to corrosive attack by diluted solutions of pure sulfuric acid at low temperatures. At 32°F (0°C), non-alloyed titanium is resistant to concentrations of about 20 percent sulfuric acid. This decreases to about 5 percent acid at room temperature. Grade 7 is resistant to about 45 percent acid at room temperature. In boiling sulfuric acid, non-alloyed titanium will show high corrosion rates in solutions with as little as 0.5 percent sulfuric acid. Grade 12 has useful resistance up to about 1 percent boiling acid. Grade 7 is useful in boiling sulfuric acid to about 7 percent concentration. The grade 5 alloy has somewhat less resistance than non-alloyed titanium. The presence of certain multi-valent metal ions or oxidizing agents in sulfuric acid inhibit the corrosion of titanium in a manner similar to hydrochloric acid. For instance, cupric and ferric ions inhibit the corrosion of non-alloyed titanium in 20 percent sulfuric acid. Oxidizing agents, such as nitric acid, chromic acid, and chlorine are also effective inhibitors.Phosphoric Acid
Non-alloyed titanium is resistant to naturally aerated pure solutions of phosphoric acid up to 30 percent concentration at room temperature. This resistance extends to about 10 percent pure acid at 140°F (60°C) and 2 percent acid at 212°F (100°C). Boiling solutions significantly accelerate attack. Grade 7 offers significantly improved resistance. At room temperature, 140°F (60°C), and boiling, grade 7 will resist concentrations of about 80, 15, and 6 percent respectively. Grade 12 offers better resistance to phosphoric acid than non-alloyed titanium but not as good as grade 7. The presence of multi-valent metal ions, such as ferric, cupric, or other oxidizing species can be used to inhibit titanium corrosion in phosphoric acid.Hydrofluoric Acid
Titanium is rapidly attacked by hydrofluoric acid of even minor diluted concentrations. It is therefore not recommended for use with hydrofluoric acid solutions or in fluoride containing solutions below pH 7. Certain complexing metal ions (i.e., Al+3, Cr+6) may effectively inhibit corrosion in diluted fluoride solutions.Sulfurous Acid
Corrosion of non-alloyed titanium in sulfurous acid is low: 0.02 milli inches per year - mpy (0.0005 mm/y) in 6 percent concentration at room temperature. Samples exposed to sulfurous acid (6 percent sulfur dioxide content) at 212°F (100°C) showed a corrosion rate of 0.04 - mpy (0.001 mm/y).Other Inorganic Acids
Titanium offers excellent resistance to corrosion by several other inorganic acids. It is not significantly attacked by boiling 10 percent solutions of boric or hydriodic acids. At room temperature, low corrosion rates are obtained on exposure to 50 percent hydriodic and 40 percent hydrobromic acid solutions.Mixed Acids
The addition of nitric acid to hydrochloric or sulfuric acids significantly reduces corrosion rates. Titanium is essentially immune to corrosion by aqua regia (3 parts HCl: 1 part HNO3) at room temperature. Grades 2, 7 and 12 show respectable corrosion rates in boiling aqua regia. Corrosion rates in mixed acids will generally rise with reductions in acid component concentrations or temperature.CORROSION OF TITANIUM IN AMBIENT SEAWATER
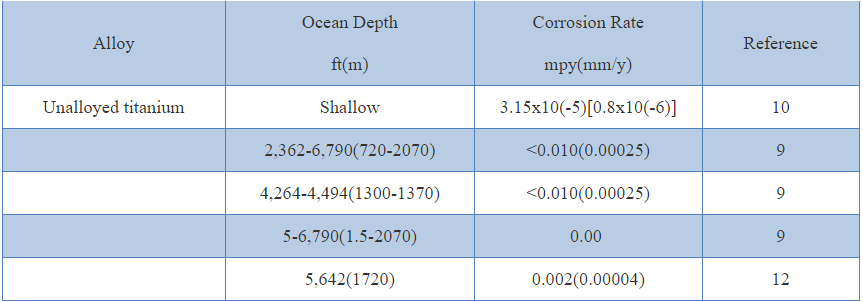
Erosion of unalloyed titanium in seawater containing suspended solids
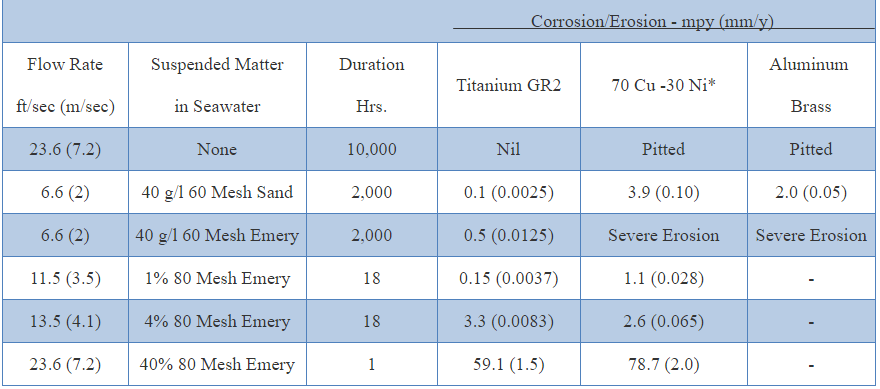
Erosion of unalloyed titanium in seawater Locations